Chetan Rupakheti, Rachael Al-Saadon, Yuqi Zhang, Aaron M. Virshup, Peng Zhang, Weitao Yang, and David N. Beratan (2016)
Contributed by Jan Jensen
Contributed by Jan Jensen
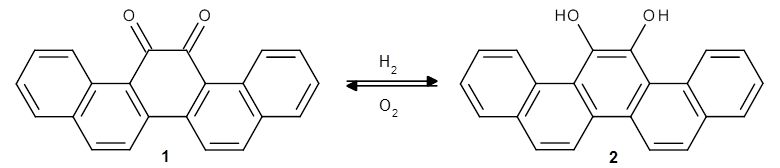
This paper uses the property-optimizing ACSESS (PO-ACSESS) method to create a set of diverse set of candidates for organic light-emitting diodes (OLEDs) with efficient blue emissions. There are over 10$^{60}$ synthetically feasible, low molecular eight organic molecules so it is impossible to do an exhaustive search of this so-called small molecule universe looking for better blue OLEDs. Yang and Beratan have therefore developed PO-ACSESS, a stochastic genetic algorithm to extract a set of diverse molecules with a given set of properties.
PO-ACSESS starts with a set of seed molecules (in this case organic molecules that are known blue emitters) that are then randomly mutated and mated. A mutation may be any of a set of elementary steps such a change in bond order, ring formation, atom addition/deletion, etc. and mating is done by randomly fragmenting the molecules at a rotateable bond followed by re-combination of fragments from different parents. The new molecules that do not meet the desired stability, synthetic-feasibility, and electronic criteria are removed. To enforce diversity, molecules that are too close in chemical space are also removed and the entire process is repeated starting with these molecules. The chemical space distance between two compounds is defined as the distance between compounds based on their descriptors, such as atomic number, Gasteiger−Marsili partial charge, atomic polarizability, and topological steric index.
In the case of blue OLEDs the electronic criteria are a vertical singlet excitation energy in the 2.4−4.1 eV range (blue region), an oscillator strength of ≥0.4, and a singlet-triplet gap ≤ 0.3 eV. “Initially, the threshold is set to a less stringent value … to ensure that the population does not collapse to zero, because of the fitness constraint.” The electronic properties were calculated using semiempirical methods such as AM1, ZINDO/S, and DFTB.
The design process was done in two stages: First PO-ACSESS was used to identify 195 molecules with the desired vertical singlet excitation energies and oscillator strengths. I could not find any details on what seed molecules were used nor the number of iterations. These 195 molecules where then used as seeds for further optimization of the singlet-triplet gap. This PO-ACSESS search was run for 50 iterations where ∼90 molecules were generated per iteration that satisfied the required singlet-triplet gap constraint, which was completed in 7 days on a 16-core CPU. I’d be very curious to know how many molecule were actually screened as part of the process. Anyway, ∼60 structures with a computed singlet-triplet gap-value of ∼0.3 eV that are predicted to emit in the blue region and to have reasonably high oscillator strengths.

This work is licensed under a Creative Commons Attribution 4.0